Strange Salt
January 15, 2014
Sodium chloride (table salt,
NaCl) is a
chemical compound that appears in the very early chapters of nearly every
chemistry textbook. Sodium, which has atomic number 11, and
chlorine, which has
atomic number 17, appear very early in the
Periodic Table, in the
third period. As a
group 1 metal, sodium has a single
positive charge when it's
ionized by losing an
electron (Na
+). As a
group 17 halogen, chlorine accepts an electron to become an ion with a single
negative charge (Cl
-).
When these ions get together, it's easy to see how the stable compound, NaCl, will form. The
crystal form of NaCl is called
halite, and that's how the halogens ("salt makers") get their name. The
crystal structure of halite is the aptly named
rock salt structure, which is a
face-centered cubic crystal (see figure).
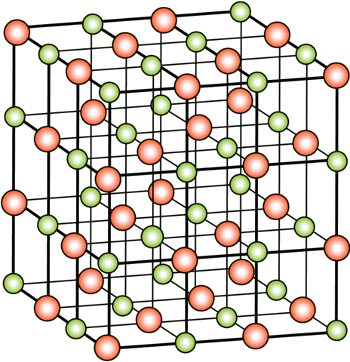
Crystal structure of rock salt (NaCl, halite)
(Image by H. Hoffmeister, via Wikimedia Commons.)
Quantum mechanics has illuminated the
atomic nature of the
chemical elements, so we now know that the
electron configuration of sodium is [Ne] 3s
1, and the electron configuration of chlorine is [Ne] 3s
2 3p
5. In forming NaCl, sodium loses its electron to become more like the stable noble gas,
neon, while chlorine accepts an electron to be more like the stable noble gas,
argon. What could be simpler, and that's the stuff of which textbook examples are made.
The reason most things happen is because
energy is minimized. In the case of halite, the sodium and chlorine atoms form this crystal structure because it's an arrangement with low
energy. That might be true for
normal temperature and pressure conditions, but sometimes putting on the
pressure will cause a
transformation from one crystal structure to another.
One example of this is
ice. At the very low temperature of 100
K and
atmospheric pressure (100
kPa), ice exists in its I
C crystalline form. Ramping up the pressure causes a transformation of crystal structures through the famous
ice-nine (IX),
II,
XV,
VIII,
X and finally
XI. Each transformation happens because the change lowers the crystal energy.
Increased pressure often has a beneficial affect on useful physical properties, such as
superconductivity. When it's found that pressure increases the superconducting
critical temperature of a particular superconductor, the material is doped with large atoms to simulate the pressure effect at atmospheric pressure. The transition temperatures of many
high temperature superconductors are routinely increased using this technique.[1]
A large international group of
scientists from
China, the
United States,
Russia, and
Germany have now demonstrated that pressure allows formation of unexpected compounds of sodium and chloride that violate the presumed charge-pairing rule for
ionic compounds.[2-6] The team was led by
Artem Oganov of
Stony Brook University and
Alexander Goncharov of the
Carnegie Institution.[5-6] Compounds with the
stoichiometries, Na
3Cl and NaCl
3, were proven to exist at high pressure by
X-ray measurements.[5-6]
The "high pressures" used weren't all that high. The compounds were demonstrated at about 200,000
atmospheres, which is far less than the 3.6 million atmospheres at
Earth's center.[5-6] Pressure studies of NaCl itself have demonstrated a transition from rock salt to the B2 phase, a cubic,
eight-fold coordinated phase, at about 30
GPa.[3] Complete
metallization of NaCl is predicted to occur above about 300 GPa, but no sodium-chlorine compounds other than NaCl were known before these experiments.[3]
As happens more often with today's better
computation tools,
theory led
experiment. Crystal structure prediction algorithms predicted the existence of Na
3Cl, Na
2Cl, Na
3Cl
2, NaCl
3, and NaCl
7.[2] Cubic and
orthorhombic NaCl
3, and two-dimensional metallic
tetragonal Na
3Cl, were formed under pressure to test the predictions. Although these compounds violated the traditional rules of chemistry, they were found to be
thermodynamically stable under the high pressure condition.[2]
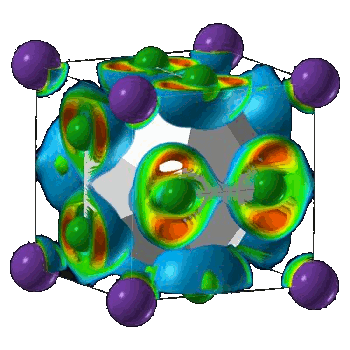
Crystal structure of NaCl3.
(Illustration by Artem Oganov, from Carnegie Institution press release.)
The lead author of the paper,
Weiwei Zhang, a
visiting scholar at Stony Brook, summarized how these results deviate from classical chemistry.
"These compounds are thermodynamically stable and once made, remain so indefinitely... Classical chemistry forbids their very existence. Classical chemistry also says atoms try to fulfill the octet rule - elements gain or lose electrons to attain an electron configuration of the nearest noble gas, with complete outer electron shells that make them very stable. Well, here that rule is not satisfied."[5-6]
Alexander Goncharov of the Carnegie Institution sees the possibility for future work.
"If this simple system is capable of turning into such a diverse array of compounds under high-pressure conditions, then others likely are, too... This could help answer outstanding questions about early planet cores, as well as to create new materials with practical uses."[4]
This work was supported in the US by the
National Science Foundation, the
Army Research Office and
DARPA, among other institutions.[4]
References:
- J. S. Schilling, "What High Pressure Studies Have Taught Us About High-Temperature Superconductivity," Frontiers of High Pressure Research II: Application of High Pressure to Low-Dimensional Novel Electronic Materials (Kluwer Academic Publishers, 2001), pp. 345-360 (PDF file).
- Weiwei Zhang, Artem R. Oganov, Alexander F. Goncharov, Qiang Zhu, Salah Eddine Boulfelfel, Andriy O. Lyakhov, Elissaios Stavrou, Maddury Somayazulu, Vitali B. Prakapenka and Zuzana Konôpková, "Unexpected Stable Stoichiometries of Sodium Chlorides," Science, vol. 342, no. 6165 (December 20, 2013), pp. 1502-1505.
- Jordi Ibáñez Insa, Perspective - Geochemistry-Reformulating Table Salt Under Pressure, Science, vol. 342, no. 6165 (December 20, 2013), pp. 1459-1460.
- Throwing out the textbook: salt surprises chemists, Carnegie Science Press Release, December 19, 2013.
- Salzige Überraschung - Forscher finden "verbotene" Verbindungen von gewöhnlichem Kochsalz, Deutsches Elektronen-Synchrotron Press Release, December 19, 2013 (in German).
- Salty surprise -- ordinary table salt turns into 'forbidden' forms, Deutsches Elektronen-Synchrotron Press Release, December 19, 2013 (in English).
Permanent Link to this article
Linked Keywords: Sodium chloride; Na; Cl; chemical compound; chemistry; textbook; sodium; chlorine; atomic number; Periodic Table; third period; group 1; alkali metal; positive charge; ionization; ionized; electron; group 17 halogen; negative charge; crystal; halite; crystal structure; rock salt structure; face-centered cubic crystal; Wikimedia Commons; quantum mechanics; atomic orbital; chemical element; electron configuration; neon; argon; energy minimization; energy; standard conditions for temperature and pressure; normal temperature and pressure condition; pressure; phase transition; transformation; ice; kelvin; K; atmospheric pressure; pascal; kPa; ice-nine; ice II; ice XV; ice VIII; ice X; ice XI; superconductivity; superconducting phase transition; critical temperature; high-temperature superconductivity; high temperature superconductor; scientist; China; United States; Russia; Germany; ionic compound; Artem Oganov; Stony Brook University; Alexander Goncharov; Carnegie Institution for Science; stoichiometry; X-ray crystallography; atmosphere; Earth's center; eight-fold coordinated; GPa; metallization; computer simulation; computation tool; theory; experiment; orthorhombic crystal system; tetragonal crystal system; thermodynamics; thermodynamically; Weiwei Zhang; visiting scholar; octet rule; planetary core; material; National Science Foundation; Army Research Office; DARPA.